 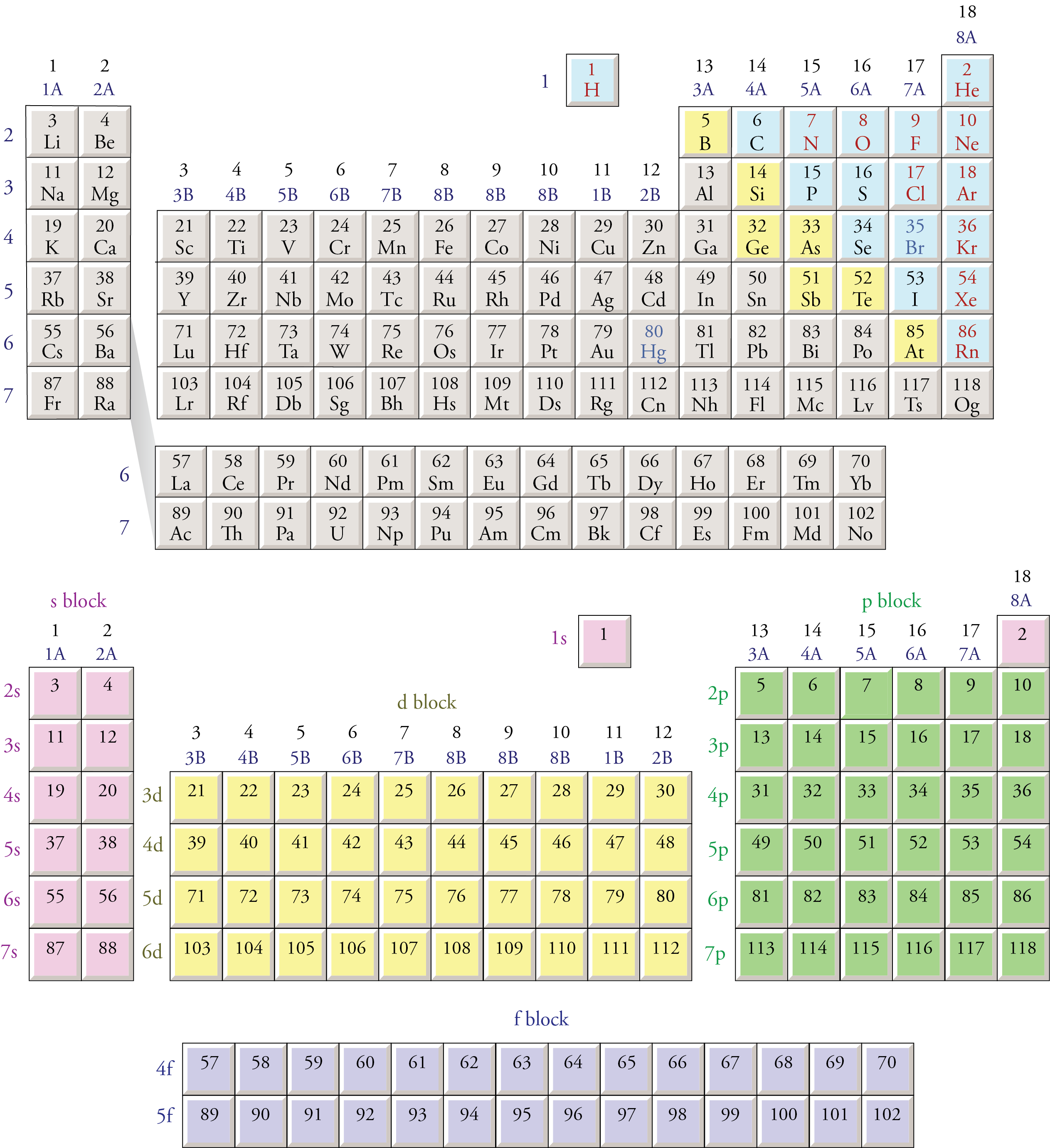
Many workers were occupationally exposed to beryllium as part of the nuclear reactor and weapons industry in the U.S. Because of the extreme inhalation hazard of Be, allowable atmospheric levels are very low. This is because of its adverse health effects, including berylliosis, a disease marked by lung deterioration. In a sense, beryllium is somewhat the opposite of a green element. Beryllium has found widespread application in aircraft brake components where its very high melting temperature (about 1290˚C) and good heat absorption and conduction properties are very advantageous. 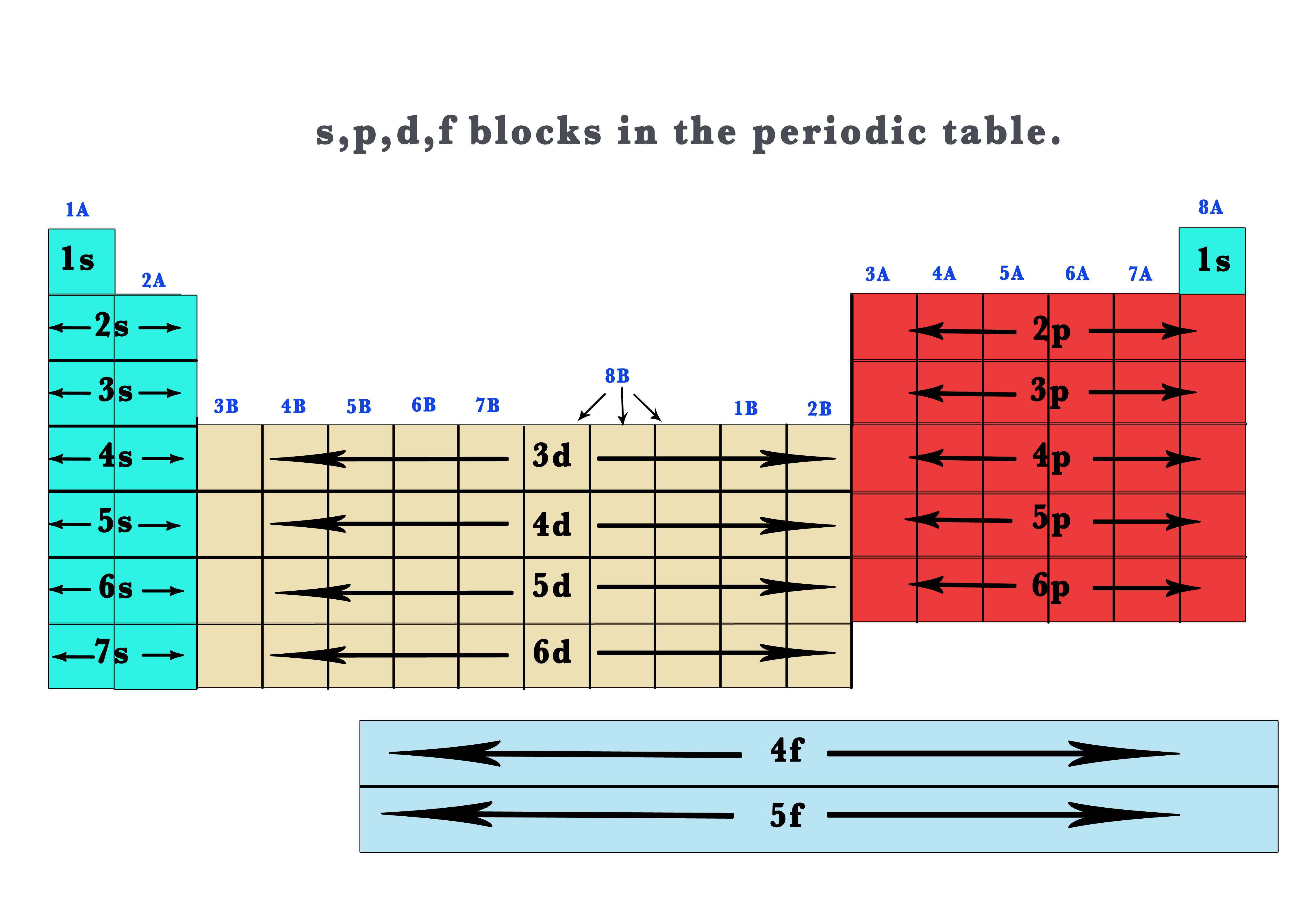
Among the devices for which beryllium alloys are especially useful are various specialty springs, switches, and small electrical contacts. Beryllium alloys can be blended that are good electrical conductors and that are nonsparking when struck, an important characteristic in applications around flammable vapors. Melted together with other metals, a process that produces alloys, beryllium yields metal products that are hard and corrosion-resistant. Since the beryllium atom needs to lose 2 electrons to reach the two-electron helium electron configuration, it produces doubly charged Be2+ cations.īeryllium has some important uses in metallurgy. When the beryllium atom is oxidized to form a beryllium cation, the reaction isīe: → Be2+ + 2e- (lost to another atom) (3.5.1) In addition to 4 protons in their nuclei, beryllium atoms also have 5 neutrons. Beryllium also has 2 outer shell electrons, so its Lewis symbol is Like atoms of all the elements in the second period of the periodic table, beryllium, atomic number 4, atomic mass 9.012, has 2 inner shell electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
